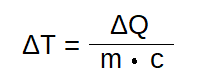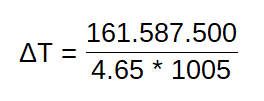Based on the current studies and discussion, since many years, the co2 – carbon dioxyd – level will raise on because fireing fossil fuels. Thats not new to us. Co2 prevents emitting of leaving radiations during back-reflections to the ground (…earth). There are more oxyds, reacting more or less stronger or weaker, like e.g. methan ch4. And its very critical if the co2 value goes against 400 ppm – parts of co2 per million. That means, that a tiny value of 0,04% and more leads to a higher temperature and advanged the greenhouse effect.
A common process: transfer co2
We know, thats a common process. The co2 is taken by components in fauna, like leafs. The „c“ of co2 is going to matter „carbon“, wood, something, and the o2 is passing the leaf as new clean air „o2“. This example and other, more complexe transfers in weather and oceans care for a level with 200 to 300 ppm in a balance. But through taken and fireing up old carbon and oil from the grounds, we increase the ppm up to 413 (Source: https://en.wikipedia.org/wiki/Keeling_Curve, 2013)
Complex system of transferation warm air to the space
We live in a natural circle of increasing and declining temperatures. On the day-side of the earth, the sun warms up earth and hours later, the warmed up earth emits the the space. Prof. Schellnhuber describs this mechanism in his book „Selbstverbrennung“, published in year 2015 (page 69ff) like a complex mechanism, in which lower athmospheric layer give because of saturation the warmness up to higher shifts until it is transfered to the space.
But there are other drivers than co2? Hows about this…
Hows about the following adoption: we get cole and gas from the inside of our earth – which was never planned for burning up again – and fire it up. And so it gets warmer… remember: the main failure, you can believe is, that energy (no matter what or where the source is) can not be consumed, i repeat: CAN NOT BE CONSUMED. Energy can only be transfered from one condition to an other condition. You fire up gas: you transfer matter from chemical, liquid state to radiant head energy „warmess“. Based on this complicated (yes, if you think about, its getting very complicated) process of transformation, i will describe a well-known application: the car with a combustion engine. In german-speaking countries, we know the word „combustor“ – in german „Verbrenner“.
The basics of combusting fuel in a simple car
In all modern cars (yes, lorries, trucks, and so on) you have a system, which is detecting if you are driving „economical“ enough. It checks everytime, if your engine is firing up fuel with the „best“ value of air. This device is called the „oxygen sensor“ or „lambda sensor“.

The lambda sensor checks up the parts of oxygen outside the car and inside the exhaust pipe and the relation of fuel and oxygen. You are driving good, it means that your mix with fuel and air is good, if the value of lambda is simply „1“. It means again, that your engine is needing a relation of 14 to 1. For each 1 kg of Fuel, you need about 14 kg of more or less clean air. In some cases, a lower or higher value of air is possible, depends on several scenaries. The sensor can give a power signal to a controller which correct values of fuel and air so the combuster works with perfect performances. But at all, you need 14 kg of air for 1 kg fireing up fuel. And another important thing is, that the optimal working temperature of your lambda sensor is around 800 °C. Remember this: this stuff in your exhaust pipe does have a temperature of about 800°C.
Think further: the consumption of fuel in a car
Nowadays, common cars in the western hemisphere need between 4 and 10 Liters of fuel. Let us take a value of 6 Liters per 100 km. Btw: i know, real us cars will need 6 Liters for starting up the engine and driving through the town. 🙂 Muscle Cars and SUVs (in europe, too) will need a lot of more. But lets start with a common car.
Think further: how much is 1 kg of fuel?
If we think about this stuff, you need to know: thats the matter of thermo dynamics, a wide field in science. But we need just a little of it. Because of fuel is lighter than water, 1 Liter does mean 0.775 kg and 1 kg does make 1.29 Liter. We need this values later.
1 kg of fuel does have chemical energy, can be convertet to a other form of energy. For our example, we convert 1 kg of fuel from chemical energy in thermal energy, a well known form.
1 Liter (means 0.775 kg) of fuel does have 34.7 Megajoule[MJ] of energy . With this formula, you can calculate the head difference.
Delta T is the temperature difference, the result by devide out Delta Q by the product of m and c . It means, if you set up a lot of enery (Delta Q in Joule [J]) to a material with a specific weight (the mass m in kilogram [kg]) and with a effective heat capacity (c in J/kg), you will get the difference in temperature Kelvin (K). Simple you can say you set up energy in Joule against mass of material and inertia (… the „Trägheit“). And as a result, you will get the delta – the difference – between the old and new temperature.
Lets take the example with water. If you have a swimming pool with a size of 3 meter to 5 meter and a depth of 2 meter, you will get 35.000 liters or kilogram of water (kg and liter of water are the same, thats the m). Water does have a effective heat capacity of 4184 J/kgK (=c). And if you put in a energy of 1 Liter of fuel, that means 34.000.000 Joule (Delta Q), so you will get a result of Delta T = 0,237 ° Kelvin. If the pool water had a temperature of 20.0 °C, now it will have 20.237 °C – in theory.
If we go further with our example above of 6 Liters of fuel, we will get 34 MJoule * 6 = 204.000.000 Joule [Delta Q]. The numerator in our formula is raising up and we will get a plus of 1.423 °C – in theory. It means, that with a value of 6, you can drive about 100 km or heat up a pool up to 1.423 °C.
Think further: heat up air with 6 Liters of fuel.
Lets take a other example with air. If we go on with it, we need to know, that 1 cubic meter of air (1m * 1m * 1m) does have a weight of 1.25 kg – so we have the mass m. The effective heat capacity of air is 1005 J/kgK – so we have the c. By the way, c means the possibility to save heat energy. And to fire up this stuff, we take the same energy like above: 1 Liter of fuel with the power of 34.000.000 J. Now imaging: if you fire up 1 cubic meter of air with 1 Liter of fuel, you will get a delta of 28.814 °C – in theory. And if we put in 6 liters of fuel, we have a heat box of 172.885 °C – in theory.
Now we take a view to our lambda sensor. If the sensor meassure up 1 kg of fuel (remember above: thats 1.29 Liter) to 14 kg of air, you can say that 6 Liters of fuel are 4.65 kg. And if 1.25 kg is 1 cubic meter, it means that 14 kg are a volume like 11.2 cubic meter of air. Imaging: 11.2 cubic meter is the same like a room in the size about 3 m * 2 m and 2 m in height. This value of air you need to fire it to drive about 100 km. And awsome is the temperature.
If we take our formula above, we have a energy power of 4.65 kg fuel with 34,7 MJ * 4.65 = 161.587.000 Joule (the Delta Q). This power we set against the air’s effecive heat capacity of 1005 J/kgK (= c) and the mass of air 4.65 kg. After this we get a Delta in Temperature of 1.429,18 °C.
That means: if we heat up about 12 cubic meter of air with 6 Liters of fuel, the temperaure raises up Delta T = 1.429,18 °C – in theory.
Think further: how much is 100 km you drive with 6 Liters of fuel?
The distance of 100 km is like you drive a route of 30 km to work and back with a shopping stop in the neighbour town, or what else. Day by day. Every time you start your engine and do a road trip with 100 km, you heat up air.
Think further: there was never a plan to fire up fossil fuels.
As i mentioned above. There was never a plan to burry out old fossil fuels and fire it up. But thats what we doing day by day since about 100 years.
Maybe you recognize, that i wrote more times „in theory“. Yes, that ar calculated values based on science’s formulars. But they dont lie. We know the effective heat capacity of air and the mass of air. Against this product, we set the power in Joule. Thats quite simple.
Just imagine this… if you put your car in a well isolated glass room with which you can drive – yes thats quite creative – and from which no one degree for power disipation can go out or be lost: there should be after 100 km a temperature like calculated above.
After you put out your engine, all the heat of it (remember the 800°C working temperatur of the lambda sensor – thats the working temperature of your engine) can not be consumed. It can only transfered from one chemical status to a second chemical status and that means for the car: heat energy. And the dissipation factor means that heat is going to spread out them self to the environment. Your car would have a temperator on 800°C, it would glow in a right red color!
The main message, i want give to you:
- energy can not be consumed; it can only be transfered
- e.g. from chemical energy like fuel to heat energy like warmness
- a consumption engine is like a heater with a car you can drive
- driving 100 km is like the same as heating up a room to 1.429 °C.
Now, decide for your self. Is co2 the only warming driver or is there a possibility to think about higher temperatures by fireing up fuel and diesel…
Think further…